
science chemistry miscibility Fundamental Photographs The Art of
This answer is: Add your answer: Earn + 20 pts Q: Is ch3ch2oh polar or nonpolar Write your answer. Submit Still have questions? Find more answers Ask your question Continue Learning about.

draw the structure of ethanol molecule toctocquienes
CH 3 CH 2 OH represents the chemical formula for ethanol, aka ethyl alcohol, a primary alcohol commonly found in alcoholic beverages, cosmetics, perfumes, and spirits. Ethyl alcohol is a clear, colorless liquid (molar mass = 46.07 g/mol) often used as a solvent for various industrial applications.

is ch3nh2 polar
Several intermolecular bonds exist and these include: 1) Hydrogen bonds - the strongest; formed between polar molecules 2) London dispersion forces (LDF) - the weakest; formed between nonpolar molecules 3) Ion-dipole bonds - formed between polar and ionic compounds Personally CH4 should not be included in the list because it is naturally in the.

Is H2O polar or nonpolar and why? YouTube
Polar vs Non-Polar molecules . As indicated in Table 2.6, the nature of molecular polarity determines the types of force(s) applied to a certain substance. So here we will have discussions about how to tell whether a molecule is polar or non-polar. The polarity of the compound can be determined by its formula and shape.

Is oxygen gas polar or nonpolar? Gek Buzz
Is the molecule C H 3 C H 2 O H polar or nonpolar? Explain. Polar and Non-Polar Molecules The electronegative difference between the atoms gives rise to polarity in the molecule. An example.

Is CH3Cl Polar or Nonpolar? YouTube
Helium is nonpolar and by far the lightest, so it should have the lowest boiling point. Argon and N 2 O have very similar molar masses (40 and 44 g/mol, respectively), but N 2 O is polar while Ar is not. Consequently, N 2 O should have a higher boiling point. A C 60 molecule is nonpolar, but its molar mass is 720 g/mol, much greater than that.

SOLVED CH3CH2OH has a much higher boiling * 3 points point than CH3O
5. "Borderline" Polar Aprotic Solvents Have Small Dipole Moments And Low (<10) Dielectric Constants. These solvents have moderately higher dielectric constants than the nonpolar solvents (between 5 and 20). Since they have intermediate polarity they are good "general purpose" solvents for a wide range of reactions.

Solved Name Lab Partner Lewis Structure (ethanol,
Chemistry Chemistry questions and answers 4. Compound: CH3CH2OH (Ethanol) Lewis Structure 3-D Structure Skeletal Structure Functional Group (s) Present: Central Atom (s) Geometry: Polar or Non-Polar 5.
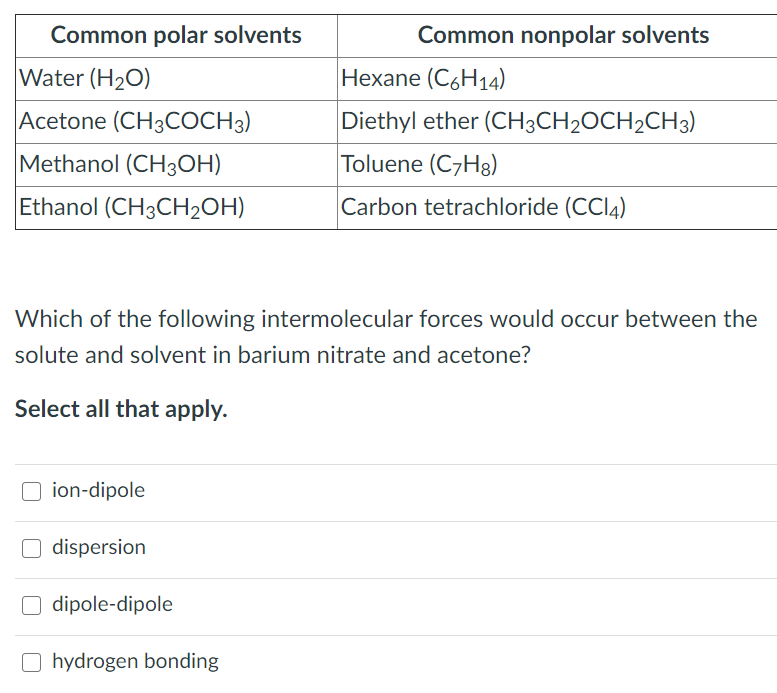
Solved Common polar solvents Water (H20) Acetone (CH3COCH3)
Answer: C2H6 (ethane) is a nonpolar molecule because it contains only nonpolar covalent bonds (C-H) bonds with both parts of the molecule cancelling out any small charge to ensure that there is no dipole moment. Is the word c2f2 polar or nonpolar? Home › Polar or Nonpolar › Is C2F2 ( Ethyne ) polar or nonpolar? Is C2F2 ( Ethyne ) polar or nonpolar?

SOLVED Draw the bondline formulas for the following compounds
The bond polarity between two atoms can be estimated if you know the electronegativity of both elements. Calculate the electronegativity difference (ΔEN) and average ( EN) of the two electronegativities, and use the table below to determine the bond type and polarity.

Best Overview Is CH3F Polar or Nonpolar Science Education and Tutorials
The molecule is symmetric. The two oxygen atoms pull on the electrons by exactly the same amount. Propane is nonpolar, because it is symmetric, with H atoms bonded to every side around the central atoms and no unshared pairs of electrons. Exercise 4.12. 1. Label each of the following as polar or nonpolar.

POCl3 Lewis Structure (Phosphoryl Chloride) Lewis, Math, Molecules
Chemistry can be a fascinating subject, especially when it comes to understanding the properties and behavior of different molecules. One such molecule that has been the subject of much discussion is CH3CH2OH. But the question is, is CH3CH2OH polar or nonpolar? Let's take a closer look to find out.Understanding Polar and Nonpolar MoleculesBefore we dive. Helium is nonpolar and by far the lightest, so it should have the lowest boiling point. Argon and N 2 O have very similar molar masses (40 and 44 g/mol, respectively), but N 2 O is polar while Ar is not. Consequently, N 2 O should have a higher boiling point. A C 60 molecule is nonpolar, but its molar mass is 720 g/mol, much greater than that. Learn to determine if CH3OH (Methanol) is polar or non-polar based on the Lewis Structure and the molecular geometry (shape).We start with the Lewis Structur. Expert-verified. Polarity of ethanol: ethanol is a polar molecule ( with dipole moment= 1.69)due to presence of hydro.. Name: Lab Partner: Lewis Structure (ethanol, CH3CH2OH) Date: Sketch - indicate polar bonds C - H ( H H Polar Bonds (Yes or No)? bo nd blo H 3c are nonPc lar H-o, c-O ave Polav Molecular Geometry at Carbon Atoms Molecular. Exercise 2.12: Vitamins can be classified as water-soluble or fat-soluble (consider fat to be a very non-polar, hydrophobic 'solvent'. Decide on a classification for each of the vitamins shown below. Exercise 2.13: Both aniline and phenol are insoluble in pure water. Predict the solubility of these two compounds in 10% aqueous hydrochloric acid.Best Explanation CH2Cl2 polar or nonpolar [N01] Science Education

Is CH3CH2OH Polar or Nonpolar? Polarity of Ethanol

Is Ch3Ch2Oh Polar Or Nonpolar? Understanding The Nature Of This
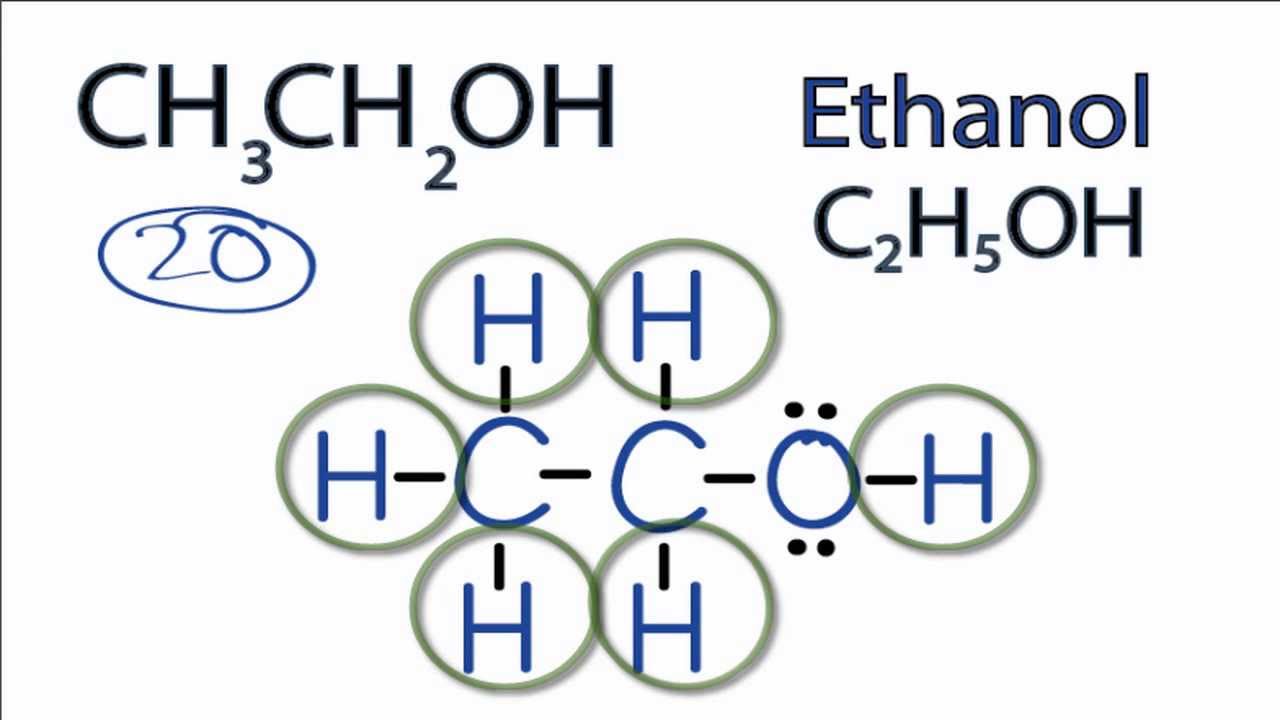
Lewis Structure For Ethanol